Welcome to Matrix Education
To ensure we are showing you the most relevant content, please select your location below.
Select a year to see courses
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Select a year to see available courses
Science guides to help you get ahead
Science guides to help you get ahead

In this article, we give you the history and uses for the elements that are in the Lanthanoid Series.
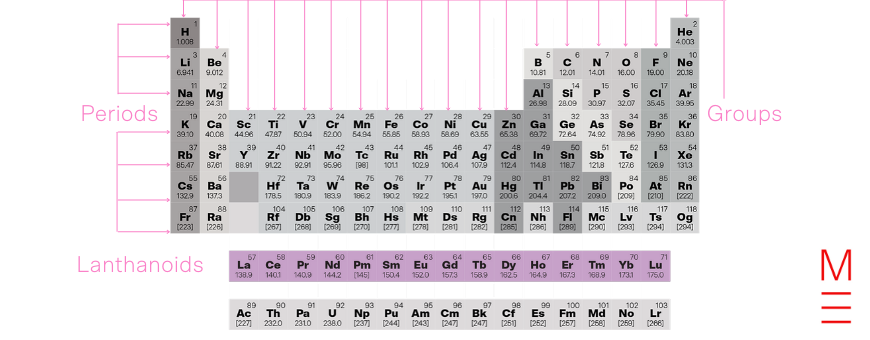
The lanthanoids, also known as the rare earths, are named after lanthanum, the first element in the series. They are defined by the progressive filling of the 4f orbital. They are all metals with similar properties and form +3 cations, as well as +2 and +4 cations in some cases. Many form colourful compounds.
Together with the actinoids, they form the f-block.

All the key Chemistry concepts and formulas you need to revise, in one foldable cheatsheet.

Fill out your details below to get this resource emailed to you.
"*" indicates required fields
Click on the following elements to learn more about them:
In 1838, following the discovery of cerium in what was believed to be its oxide, ceria, Carl Gustaf Mosander showed that ceria contained additional oxides which he named lanthana and didymia. He isolated lanthana from impure cerium nitrate and named it after the Greek lanthanein, meaning hidden, after a suggestion from Jons Jakob Berzelius. He went on to obtain impure metallic lanthanum. Lanthanum is difficult to isolate and purify, so pure samples were not produced until 1923.
Lanthanum alloys and compounds are used for parts of batteries (e.g. for electric vehicles), as pyrophoric alloys, and in few other niche applications such as in infra-red-transparent glass (called ZBLAN).
In 1803, Wilhelm Hisinger was interested in identifying the composition of a new mineral discovered in mines he owned. He and Jons Jakob Berzelius isolated a new oxide from the mineral, which indicated the presence of a new element. Berzelius named the oxide ceria after the dwarf planet Ceres that had recently been discovered, itself named after the Roman goddess of agriculture. The new element was thus named cerium, and the original mineral cerite.
Martin Heinrich Klaproth independently discovered the oxide from the same mineral in the same year and named it ockroite, in reference to its red-yellow colour, however Hisinger’s and Berzelius’s results were published first and the names they chose were accepted. In 1825, Carl Gustaf Mosander who worked with Berzelius isolated cerium metal. In the 1830s, Mosander showed that the metal was impure and was able to isolate pure cerium once he removed impurities.
Cerium is most commonly used as ceria for polishing glass. It is also used as an additive in glass to prevent darkening, in temperature-stable pigments and phosphors, and in catalytic converters.
Praseodymium and neodymium were discovered together in what was believed to be an element called didymium. In the 1830s and 40s, ceria (believed to be cerium oxide) was shown by Carl Gustaf Mosander to contain additional oxides which he named lanthana and didymia. The latter was mistaken for the oxide of a new metal named didymium, after the Greek didymos meaning twin, given its similarity to lanthanum, and given the symbol Di.
Eventually it became clear that didymium was not an element and that its properties varied. Starting from 1879, other elements were extracted from didymium, and in 1885, Carl Auer von Welsbach separated what remained as didymium into two elements – one that formed pink salts (neodymium) and one that formed green salts. He named this second element praseodymium meaning the green twin from Greek prasio, meaning leek-green.
Praseodymium was initially used to colour glass and ceramics but was mostly replaced by cheaper alternatives, except perhaps for glass used in welding goggles. It is used in rare-earth magnets along with neodymium and in some phosphors.

Praseodymium and neodymium were discovered together in what was believed to be an element called didymium. In the 1830s and 40s, ceria (believed to be cerium oxide) was shown by Carl Gustaf Mosander to contain additional oxides which he named lanthana and didymia. The latter was mistaken for the oxide of a new metal named didymium, after the Greek didymos meaning twin, given its similarity to lanthanum, and given the symbol Di.
Eventually it became clear that didymium was not an element and that its properties varied. Starting from 1879, other elements were extracted from didymium, and in 1885, Carl Auer von Welsbach separated what remained as didymium into two elements – one that formed pink salts and one that formed green salts (praseodymium). He named the first element neodymium meaning the new twin from the contraction of the Greek neos didymos.
The most common use of neodymium is in neodymium-iron-boron alloy rare earth magnets, which are the strongest permanents magnets. These are used in microphones, speakers, headphones, electric vehicle motors and some electric generators. It is also used to colour glass, and to make high power lasers.

Prior to promethium’s discovery, neodymium and samarium were believed to be consecutive elements in the periodic table. In 1902, Bohuslav Brauner realised their properties differed more than other similar consecutive metals and predicted that an unknown metal existed between them. This was supported by Henry Moseley in 1914 who showed that element 61 was yet to be discovered. In 1926, two groups claimed to have discovered it, one group from Florence naming it florentium, and another from Illinois, naming it illinium, however in both cases the scientists had misidentified other known elements. In 1934, Josef Mattauch showed that element 61 would form no stable isotopes and may thus not occur naturally on Earth.
In 1938, a nuclear experiment produced an element which was neither neodymium or samarium. It was inferred to be element 61 and named cyclonium, however this discovery is generally overlooked due to a lack of evidence. Finally, in 1945, Jacob Marinsky, Lawrence Glendenin, and Charles Coryell identified element 61 in the fission products of uranium fuel from a nuclear reactor. They proposed the name clintonium, after the Clinton Laboratories where they worked. Grace Coryell (wife of Charles) suggested the name prometheum, after the Titan Prometheus in Greek mythology who stole fire from the gods and granted it to humans, a reference to how it was obtained. The name was modified with the -ium suffix used to name metals.
Promethium has virtually no applications, given it has no stable isotopes (longest half-life is 17.7 years). One exception is its use in luminous paint.
The discovery of samarium is credited to Paul Emile Lecoq de Boisbaudran, who isolated samarium compounds in 1879 from the mineral samarskite, and identified the new element through its absorption spectrum. He named the metal samaria after the samarskite, which was in turn named after Vassili Samarsky-Bykhovets, a Russian official who had originally given access to minerals from the Urals in Russia to be studied, which had lead to the discovery of samarskite. The name was changed to samarium to incorporate the -ium suffix used for metals, and although the symbols Sa and Sm were both used, Sm was adopted.
The main use of samarium is in samarium-cobalt magnets. Whilst not as strong as neodymium magnets they remain stable at higher temperatures and have a higher resistance to demagnetisation.

Europium was discovered by either Paul Emile Lecoq de Boisbaudran or Eugène-Anatole Demarçay, though the latter is usually credited. In 1892, Boisbaudran investigated samarium-gadolinium compounds and identified spectral lines in them that belonged to neither element, indicating the presence of a new element. In 1896, Demarcay identified the new element as an impurity in samarium (which Boisbaudran had discovered) and isolated it in 1901. Demarcay named the element europium, after Europe.
Europium oxides are used as phosphors, previously in TV screens, and currently in fluorescent lights.

In 1880, Jean Charles Galissard de Marignac identified new spectral lines in the minerals gadolinite and cerite. He separated an oxide associated with these spectral lines from cerite and named it gadolinia after the mineral. New oxides were known to be attributable to new metals, in this case gadolinium. The metal, oxide and mineral are all named after chemist Johan Gadolin who had worked on the mineral almost a century earlier. Paul Emile Lecoq de Boisbaudran isolated gadolinium metal in 1886.
Gadolinium has very few niche applications such as a neutron-absorber in neutron therapy for cancer and in other settings related to nuclear reactors.
Both terbium and erbium were discovered in 1843 by Carl Gustaf Mosander. He discovered two new oxides in the mineral gadolinite – one was pink and was named terbia and the other was brown and named erbia. In 1877, the name of the brown oxide was changed to terbia, meaning the metal associated with it was named terbium. The name derives from the village of Ytterby in Sweden where gadolinite was discovered.
Some terbium compounds are used in electronic devices like sonar systems as they have specific magnetic properties. Terbium oxide is used as a phosphor, previously in TV screens, and currently in fluorescent lights.
Paul Emile Lecoq de Boisbaudran discovered a new oxide in samples of holmium oxide in 1886, and isolated a new metal from this oxide. He named the metal dysprosium after the Greek dysprositos meaning hard to get, as the isolation of the metal had taken him 30 attempts.
Dysprosium has few niche applications, which include lasers, lighting, applications in nuclear reactors and in magnets and magnetic materials.
Holmium was discovered by Jacques-Louis Soret and Marc Delafontaine in 1878 through spectroscopic techniques, and named element X. It was independently discovered in 1879 by Per Teodor Cleve as one of two impurities in erbia (erbium oxide). One of these was a brown oxide he named holmia after the Latin name for his home town of Stockholm, thus the metal associated with it was named holmium. (The other was green thulium oxide.) Holmium was originally mistaken to be element 66 as early samples tested were contaminated with dysprosium.
Holmium is used in magnetic applications, in lasers, and as a neutron absorber in nuclear reactors.

Both terbium and erbium were discovered in 1843 by Carl Gustaf Mosander. He discovered two new oxides in the mineral gadolinite – one was pink and was named terbia and the other was brown and named erbia. In 1860, the name of the pink oxide was changed to erbia, and the metal associated with it was named erbium. The name derives from the village of Ytterby in Sweden where gadolinite was discovered.
Erbium is used to give glass a pink colour, and is used in optical amplifiers in optical fibre telecommunications networks and in lasers.

Thulium was discovered in 1879 by Per Teodor Cleve as one of two impurities in erbia (erbium oxide). One impurity was the brown holmium oxide, and the other impurity was a green oxide he named thulia, after the Greek Thule, a possible reference to Scandinavia in Ancient Greek writings. The metal associated with thulia was named thulium and given the symbol Tu, which was later changed to Tm. Thulium metal was isolated in 1911 by Charles James.
Thulium is used in infrared lasers for military and medical applications, and thulium radioisotopes are used as portable radiation sources.

In 1878, Jean Charles Galissard de Marignac extracted a new substance from erbia, which had itself been extracted from the mineral gadolinite. He named the new substance ytterbia, after Ytterby in Sweden which was associated with the discovery of the gadolinite and yttrium. He proposed that the ytterbia was associated with a new element that he named ytterbium. In 1907, three chemists independently showed that ytterbia could be separated into two components, and hence indicated the existence of two elements.
Georges Urbain named the first of these elements neoytterbium, Carl Auer von Welsbach named it alderbaranium, and Charles James did not propose a name (the second element identified was lutetium). After a dispute between Urbain and von Welsbach about precedence, the Commission on Atomic Mass (of which Urbain was a member) recognised Urbain’s discovery in 1909 on the basis that it was published first. Urbain’s name of neoytterbium was thus adopted, however the name reverted to ytterbium as had been proposed by de Marignac.
Ytterbium is used in small quantities in some steels, and in infrared lasers.

In 1878, Jean Charles Galissard de Marignac extracted a new substance from erbia, which had itself been extracted from the mineral gadolinite. He named the new substance ytterbia, after Ytterby in Sweden which was associated with the discovery of the gadolinite and yttrium. He proposed that the ytterbia was associated with a new element that he named ytterbium.
In 1907, three chemists independently showed that ytterbia could be separated into two components, and hence indicated the existence of two elements. Georges Urbain named the second of these elements lutecium, Carl Auer von Welsbach named it cassiopeium, and Charles James did not propose a name (the first element was ytterbium). After a dispute between Urbain and von Welsbach about precedence, the Commission on Atomic Mass (of which Urbain was a member) recognised Urbain’s discovery in 1909 on the basis that it was published first. Urbain’s name of lutecium was accepted except in German-speaking countries which used cassiopeium.
Around 1950, the spelling was changed to lutetium and the name was accepted universally. The element is named after the Roman-era city of Lutetia, the predecessor of Paris, as Urbain was French and director of the Chemical Institute in Paris.
Due to its rarity and similarity to other lanthanides lutetium has few uses, which include catalysis and phosphors.

Matrix+ will give you access to HSC Chemistry Experts, wherever you are! Learn more.
Get ahead in HSC Chemistry
Expert teachers, comprehensive resources, 24/7 help with Ask Max! Learn from home with Matrix+ online.
© Matrix Education and www.matrix.edu.au, 2025. Unauthorised use and/or duplication of this material without express and written permission from this site’s author and/or owner is strictly prohibited. Excerpts and links may be used, provided that full and clear credit is given to Matrix Education and www.matrix.edu.au with appropriate and specific direction to the original content.