Welcome to Matrix Education
To ensure we are showing you the most relevant content, please select your location below.
Select a year to see courses
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Learn online or on-campus during the term or school holidays
Select a year to see available courses
Science guides to help you get ahead
Science guides to help you get ahead
How ready are you for the HSC? Here are the essential Chemistry techniques you must know for the HSC.

Join 75,893 students who already have a head start.
"*" indicates required fields
You might also like
Related courses

Join 8000+ students each term who already have a head start on their school academic journey.
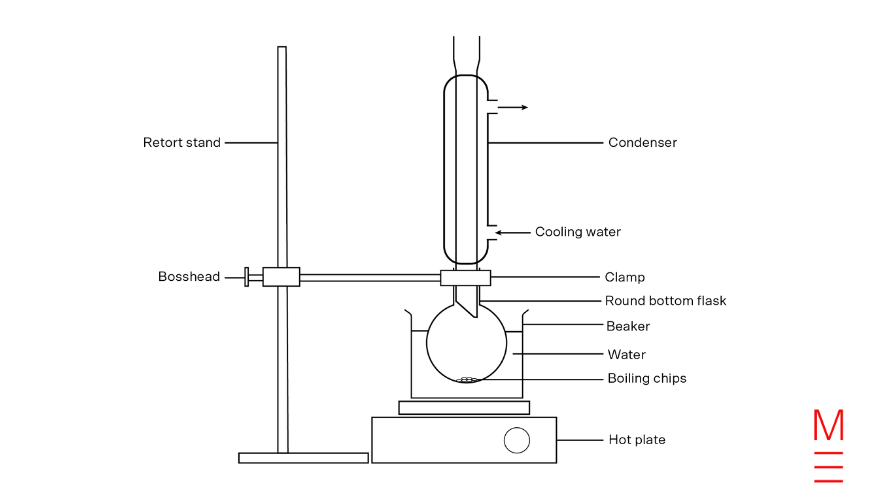
The following essential HSC Chemistry Exam Techniques have been compiled from the NESA website and the 2016 Term 1 Matrix Education Year 12 Chemistry Theory Book.
Get ahead in HSC Chemistry
Expert teachers, comprehensive resources, 24/7 help with Ask Max! Learn from home with Matrix+ online.
Written by Matrix Science Team
The Matrix Science Team are teachers and tutors with a passion for Science, across Biology, Chemistry and Physics, and a dedication to seeing Matrix Students achieving their academic goals.© Matrix Education and www.matrix.edu.au, 2025. Unauthorised use and/or duplication of this material without express and written permission from this site’s author and/or owner is strictly prohibited. Excerpts and links may be used, provided that full and clear credit is given to Matrix Education and www.matrix.edu.au with appropriate and specific direction to the original content.